What is denitrification efficiency?
Denitrification efficiency is the percentage of inorganic nitrogen (DIN) released from the sediment as dinitrogen gas (N2) during the decomposition of organic matter1:
( (N2-N / (DIN + N2-N) * 100%)
Significance of denitrification efficiency
Denitrification is one of the few processes capable of counteracting eutrophication because it can permanently remove nitrogen from the system as dinitrogen gas (Figure 1)1234. When denitrification efficiencies are lowered, more nitrogen is recycled to the water column as ammonium (Figure 1). Nitrogen that is recycled to the water column (and not denitrified) stimulates further cycles of primary production, and therefore can continue the supply of organic matter to the sediment, leading to more decomposition and more dissolved oxygen consumption, and potentially to anoxic and hypoxic events 123.
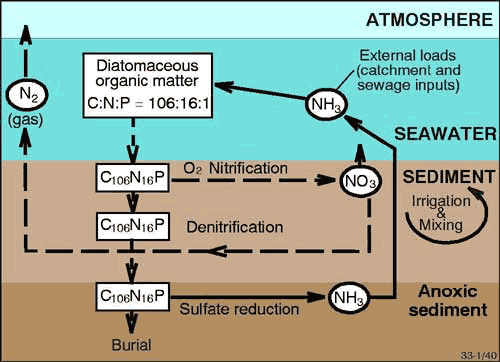
Figure 1. Schematic diagram of N cycling in sediments (Modified after Heggie et al., 19993 ).
What causes denitrification efficiency to change?
Carbon loading (a.k.a. trophic status) is perhaps the main control on the denitrification efficiencies of sediment [1-4]. Carbon loadings are reflected in the benthic carbon dioxide flux. There is evidence that denitrification efficiencies of >~68%, ~40%, 18% and 8% are indicative of oligotrophic, mesotrophic, eutrophic and hypertrophic conditions respectively, in shallow coastal waterways in southeastern Australia4.The denitrification efficiency optimum occurs between carbon loadings of about 50 and 100 g C m-2 y-1. 4 There is likely an overlap of aerobic and anaerobic respiration zones at the denitrification efficiency optimum4. This is caused by the existence of anaerobic micro-niches within the oxic zone and also by oxidised burrow structures penetrating into the anaerobic zone4. These enhance denitrification by improving both the NO3 and organic carbon suppy to the denitrifying bacteria.,Sediments tend to lose this 3-dimensional complexity at higher carbon loads4.
The lowering of denitrification efficiencies with increased carbon loadings results from either:
- the inhibition of nitrification caused by a lack of dissolved oxygen or exposure to hydrogen sulfide gas1235; or
- from an increase in the quantitative importance of dissimilatory nitrate reduction to ammonium16.
Salinity7 and water temperature8 can also have physiological impacts on denitrifying bacteria, and may be additional controls on denitrification efficiencies.
Waterways in which denitrification is best applied as an indicator
Denitrification efficiency is a good indicator of sustainable carbon loading rates in bays 234, shallow coastal lagoons 14 and wave-dominated estuaries 49, where it is almost always linked to nitrification occurring in the sediment (Figure 2a).
There is some evidence that the relationship between denitrification efficiency and trophic status described above may not hold in coastal waterways where denitrification is linked to water column nitrate110 (Figure 2b). High water column nitrate concentrations are most common in waterways with large amounts of suspended sediment that limits light penetration and therefore plant and algal biomass production. This would include most tide-dominated coastal waterways (e.g. deltas, estuaries and tidal creeks) because strong tidal currents resuspend fine sediment11.
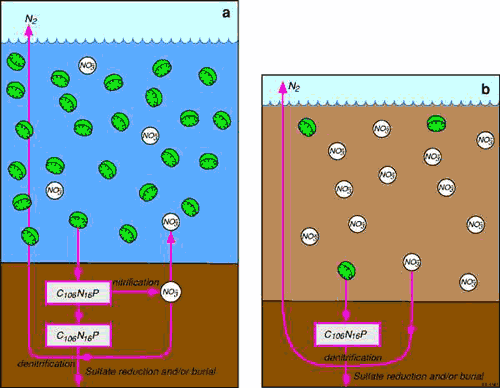
Figure 2. A comparison of denitrification in wave- (a) and tide-dominated waterways (b). Algal biomass is naturally lower in tide-dominated systems (b) than in wave-dominated waterways (a) because tidal mixing reduces the residence time of algae (green disks = diatoms) in the photic zone, and because sediment resuspended by tides increases turbidity, which reduces light available for photosynthesis12. As a consequence, the amount of nutrients that can be taken up by plants is also lower in tide-dominated waterways in comparison to wave-dominated waterways. As such, dissolved nutrient concentrations build up in the water column in tide-dominated systems, and denitrification can be coupled to nitrate in the water column rather than to nitrification occurring in the sediment. In wave-dominated systems, denitrification is more likely to be coupled to nitrification occurring in the sediment.
Considerations for measurement and interpretation
The denitrification efficiency of sediments in estuaries is determined using benthic chambers. Benthic chambers are open-bottom containers (usually constructed out of perspex) that enclose an area of sediment and overlying water. The chambers are deployed on the sediments/substratum, to capture solute movement (including nitrogen) between the sediment and the water column. The denitrification efficiency of sediments in an estuary is determined by comparing the amount of nitrogen released as a gas to the total amount of nitrogen released.
The released nitrogen gas is quantified using a Membrane Inlet Mass Spectrometry (MIMS) technique13. This involves stripping the gases from a water sample using a membrane under high vacuum. Carbon dioxide and water are removed from the gas-stream via a cold trap. The dry gas is then introduced to the mass spectrometer to measure N2/Ar ratios. The N2 concentrations are then calculated from the measured ratios using the N2/Ar calibration curves and theoretically determined argon concentrations for the sampled waters. The N2/Ar technique has several assumptions that must be met to achieve accurate rate calculations14. Different tests can be applied to ensure these assumptions are being met14.

Photo 1. A benthic chamber being deployed
In the absence of dinitrogen measurements, denitrification rates (DR) and efficiencies (%DE) can be estimated from the following equations:
DR = TDINp – DINm
DE% = =[TDINp – DINm ] * 100 / TDINp
in which [DIN]m = the measured dissolved inorganic nitrogen liberated from the sediment during the degradation of organic matter and [TDIN]p = total dissolved inorganic nitrogen flux predicted on the basis of stoichiometric relationship with the benthic carbon dioxide flux11. However, the stoichiometric approach requires that the molar proportions of carbon and nitrogen in the organic matter being degraded are Redfield, or are at least known. Problems with the stoichiometric approach can occur if one assumes that nitrogen missing from the DIN flux has been denitrified, when it may have been utilised by bacteria in sediment. This is most likely to occur in sediments with high TOC:TN ratios (>20) because microbial decomposition can be nitrogen limited under such conditions15.
Existing information and data
Major research institutions (GA, MAFRI, AIMS), universities and government (local and State) agencies gather denitrification-related data for specific research studies. Denitrification rate and efficiency data (mainly stoichiometric) for thirteen coastal waterways were compiled during the National Land and Water Resources Audit. This information is presented in summary form (e.g. medians and 25 and 75th percentiles) in the OzEstuaries database. Relationships between nutrient loads and denitrification efficiencies in different types of coastal waterways can be explored in the Simple Estuarine Response Models developed by CSIRO.
More information on nutrients (changed from natural).
Contributors
Ian Atkinson, Geoscience Australia
David Heggie, Geoscience Australia
Graham Skyring, Skyring Environment Enterprises
Arthur Webb, Southern Cross University
- Eyre, B.D. and Ferguson, A.J.P. 2002. Comparison of carbon production and decomposition, benthic nutrient fluxes and denitrification in seagrass, phytoplankton, benthic microalgae- and macroalgae-dominated warm-temperate Australian lagoons. Marine Ecology Progress Series 229, 43-59. ↩ ↩ ↩ ↩ ↩ ↩ ↩
- Berelson, W.M., Heggie, D., Longmore, A., Kilgore, T., Nickolson, G., Skyring, G., 1998. Benthic nutrient recycling in Port Phillip Bay, Australia. Estuarine and Coastal Shelf Science 56, 917-934. ↩ ↩ ↩ ↩
- Heggie, D. T., Skyring, G. W., Orchardo, J., Longmore, A. R., Nicholson, G. J., and Berelson, W M., 1999. Denitrification and denitrifying efficiencies in sediments of Port Phillip Bay: direct determinations of biogenic N2 and N-metabolite fluxes with implications for water quality. Marine Freshwater Research 50, 589-596. ↩ ↩ ↩ ↩ ↩
- Eyre, B. and A.J.P. Ferguson, 2009. Denitrification efficiency for defining critical loads of carbon in shallow coastal ecosystems. Hydrobiologia 629(1), 137-146. (DOI 10.1007/s10750-009-9765-1 ↩ ↩ ↩ ↩ ↩ ↩ ↩ ↩ ↩
- Joye, S.B. and Hollibaugh, J.T., 1995. Influence of sulfide inhibition of nitrification on nitrogen regeneration in sediments. Science 270, 623-625. ↩
- Christensen, P.B., Rysgaard, S., Sloth, N.P., Dalsgaard, T., and Schwaerter, S., 2000. Sediment mineralisation, nutrient fluxes, denitrifcation and dissimilatory nitrate reduction to ammonium in an estuarine fjord with sea cage trout farms. Aquatic Microbial Ecology 21, 73-84. ↩
- Rysgaard, S., Thastum, P., Dalsgaard, T., Bondo Christensen, P., and Sloth, N.P., 1999. Effects of salinity on NH4+ adsorption capacity, nitrification, and denitrification in Danish Estuarine sediments. Estuaries 22(1), 21-30. ↩
- Carpenter, E.J., Dunham, S., 1985. Nitrogenous nutrient uptake, primary production, and species composition of phytoplankton in the Carmans River estuary, Long Island, New York. Limnol. Oceanogr. 30, 513-526. ↩
- Fredericks, D. J. and Heggie, D. T., 2000. Are sediments a significant source of nutrients in Wilson Inlet. AGSO Professional Opinion 2000/04 Australian Geological Survey Organisation, Canberra. ↩
- Eyre, B.D. and Ferguson, A.J.P., 2002. Denitrification efficiency as an indicator of change associated with nutrient over-enrichment of shallow coastal ecosystems. Proceedings of the annual conference of the Australian Marine Sciences Association, 10-12 July 2002, Fremantle WA. ↩
- Heap, A., Bryce, S., Ryan, D., Radke, L., Smith, C., Smith, R., Harris, P. and D. Heggie., 2001. https://data.gov.au/dataset/australian-estuaries-and-coastal-waterways-a-geoscience-perspective-for-improved-and-integrated. AGSO Record 2001/07, pp. 118 ↩ ↩
- Monbet, Y. 1992. Control of phytoplankton biomass in estuaries: A comparative analysis of microtidal and macrotidal estuaries. Estuaries 15(4), 563-571. ↩
- Kana, Todd., Darkangelo, Christina., Hunt, M. Duane., Oldham, James B., Bennett, George E., and Cornwell, Jeffrey C., 1994. Membrane inlet mass spectrometer for rapid high-precision determination of N2, O2, and Ar in environmental water samples. Analytical Chemistry, American Chemical Society, United States: 66/23: 4166-4170. ↩
- Eyre, B. D., Rysgaard, S., Dalsgaard, T. and Christensen, P. B., 2002. Comparison of isotope pairing and N2/Ar methods for measuring sediment denitrification rates – assumptions, modifications and implications. Estuaries 25, 1077-1087. ↩ ↩
- Twilley, R.R., Cowan, J., Miller-Way, T., Montagna, P.A. and Mortazavi, B., 1999. Benthic nutrient fluxes in selected estuaries in the Gulf of Mexico, pp 163-209 In Bianchi, T.S., Pennock, J.R. and Twilley, R.R. (Eds), Biogeochemistry of Gulf of Mexico Estuaries, John Wiley & Sons, Inc. ↩