What are mangroves?
Mangroves comprise several species of trees and shrubs that grow along sheltered intertidal shores, mainly in tropical & subtropical coastal waterways. However, in Eastern Australia, mangroves do extend along temperate coastlines where their distribution overlaps with saltmarsh communities. In these instances, mangroves usually establish in low elevation sites where inundation is more frequent1. Australia has 39 mangrove species, which is more than half the global number23.
Mangroves can be found in all the major subclasses of coastal waterways in Australia including: tide-dominated deltas, tide-dominated estuaries, tidal creeks, wave-dominated deltas, wave-dominated estuaries and strandplains4. However, they are most common in tide-dominated waterways (e.g. deltas, estuaries and tidal creeks) and in wave-dominated deltas4.
Mangroves are adapted to the salt-water environment and to anoxic and sulfidic-rich sediments (see acid sulfate soils). For example, some are recognised by their breathing roots (i.e. pneumatophores) which obtain oxygen directly from the atmosphere when exposed at low tide (see photos 1-3). Other special features of mangroves include: buttresses and prop roots for support; salt excretion from leaf pores; and floating seedlings (viviparous propagules)2. Sheltered intertidal shorelines are also occupied by salt marshes, however saltmarshes are generally dominant in more temperate and drier regions. Where seagrass beds are found adjacent to mangroves, many material links and shared plant and animal communities exist (Photo 4)5. Changes in the distribution of mangroves have been identified as an important indicator of broader environmental change for State of the Environment reporting6.

Photo 1. Breathing roots (pneumatophores) of Avicennia marina (photo by Jon Knight, UQ) obtain oxygen directly from the air.

Photo 2. The knee roots of Bruguiera gymnorrhiza (photo CSIRO Land and Water, Environmental Remote Sensing group) provide for oxygen uptake by the mangrove roots in anoxic mangrove muds.

Photo 3. The prop roots of Rhizophora stylosa (photo Jon Knight, UQ) enable these mangroves to grow low in the intertide zone where the roots become inundated during high tides.

Photo 4. Mangroves are an integral component of the intertidal environment of Moreton Bay Qld, where they grow in conjunction with expansive mud flats and seagrass beds (foreground) (photo Jon Knight UQ).
The importance of mangroves
Mangroves offer many benefits to both natural systems and humans, and their removal has several economic consequences.
- Shoreline protection and sediment accretion; mangroves buffer the shoreline from the erosive impact of storms and waves.
- Trap and bind sediments; reducing coastal turbidity and making cleaner waters.
- Major source of primary productivity in the form of plant materials; supporting many important trophic pathways by providing a source for food chains that support many terrestrial and marine organisms2.
- Provide habitat for both marine and terrestrial organisms; a home for both plants and animals (including insects; photo 5).
- Provide nurseries for commercially important fish and prawn stocks; replenishing estuarine and coastal fisheries.
- Mangroves are a sink for atmospheric carbon; helping to reduce global carbon dioxide levels and global warming.
- Mangroves capture effluents from terrestrial runoff; providing a buffer for nutrients, metals and other toxicants entering coastal waters7.
- Source of timber and wood products; used for light construction and home energy in developing countries.
- And more recently, changes in mangroves have been proposed as a means to monitor change in coastal environments as indicators of global warming, climate change, storm effects, sea level change, pollution, and sedimentation rates.

Photo 5. Avicennia marina is the most extensive mangrove in Moreton Bay Qld. Pools like these ones are key habitats for mosquito breeding (photo by Jon Knight, UQ).
What causes mangrove areas to change?
Shoreline development (e.g. coastal urbanisation and industrialisation and conversion to aquaculture) and changes in local hydrology are the biggest threats to mangrove habitat and may cause changes in mangrove extent. Regional and global processes that influence hydrology, such as climate variability, climate change and sea level may also cause significant changes to mangrove areas. Some specific threats include:
- Storms (photo 6).
- Baffling by mangrove root systems provides a physical trap for fine sediment with loads of heavy metals8 and other toxicants. Changes in pH, redox potential (dissolved oxygen) and salinity can render these toxicants more available to keystone mangrove animals, including crabs. Increases in heavy metals can also lead to inhibition of photosynthesis and respiration in mangroves, causing die back9.
- Mangroves are susceptible to pollution10 (photo 7). Floating oil is a particular concern because it deposits on mangrove roots when the tide drops and contaminates the sediment. Mangroves die from the suffocating or toxic effects of the oil, or from the oiling of keystone burrowing animals (crabs and worms), which are essential to the health of the system11.
- Rotting drift algae from algal blooms, wrack from seagrass areas2 and excess input of sediment can smother mangrove pneumatophores causing senescence and death of trees1213.
- Phytothera introduced in landfill soils has been implicated in the die-back of mangroves14.
- Trampling can decrease the density of pneumatophores and the biomass of epiphytic algae causing a change in habitat structure14.
- Increased tidal amplitudes, caused by dredging and climate change, and changes in riverine nutrient and sediment loads, can cause the expansion of mangroves into saltmarsh areas15.
- Increased freshwater contribution from stormwater drains and regional increases in rainfall can cause the expansion of mangroves into saltmarsh areas15.
- A reduction in the duration of the wet season can lower water tables, and increase salinity stress16.
- Genetic modification of the mangrove genome due to pollution causes changes in species composition and fitness of mangroves17.
- Invasive weeds (photo 8). For example, pond apple is a weed that superficially resembles mangroves and is of particular concern because of its invasiveness, potential for spread and its environmental and economic impacts. It now infests more than 2000 ha in the Wet Tropics, and threatens melaleuca wetlands in addition to mangrove communities.

Photo 6. Die back in mangroves can result in extensive defoliation and death of mangroves, as in these Avicennia marina mangroves in Moreton Bay Qld. The most likely cause of this die back was a hail storm in 1999 (photo by Jon Knight, UQ).

Photo 7. Accumulation of town rubbish in mangrove near Tea Gardens, NSW (photo by C. Wenger)


Photo 8. Invasion of mangroves near Tea Gardens, NSW, by: (a) Lantana; and (b) Bitou bush (photos by C. Wenger).
Mangroves and Sea-level Rise
Due to the identification of regional trends of mangrove expansion, there has been considerable interest in the relationship between mangrove extent and sea-level changes associated with global warming [For example1518]. Intertidal vegetation, such as mangrove, may respond to sea-level rise by migrating upslope, or increasing their elevation through processes of vertical accretion or sedimentation so that they remain within the same tidal range19. Without such a response, mangroves suffer from a contraction in extent at the shoreline due to erosion, or submergence and death20. Since the response of mangroves to sea-level rise are numerous and vary depending on the rate and degree of sea-level rise, identifying links between changes in mangrove area and sea-level rise are difficult. By coupling analyses of change in mangrove extent with ground-based analyses of vertical accretion and surface elevation change, linkages between changes in mangrove extent and sea-level rise can be identified18.
Considerations for measurement and interpretation
Mapping changes in the distributions of mangroves is relatively straightforward, and can be undertaken by community groups such as Waterwatch. Aerial photography and satellite imagery can be used, although ground-truthing by local agencies is advised (photo 9). Identifying the causes of mangrove expansion or contraction may be difficult to identify and consideration should be given to natural forces including storm damage (see Photo 6), and changes in rainfall, tidal regime and sea level.
While mapping mangrove distribution provides considerable information about changes in extent, it rarely provides information about the condition of mangroves. Exceptions do occur when mangrove death occurs over large spatial scales, which can be observed from aerial photographs or satellite imagery13. For this reason it is advisable to accompany analyses of changes in extent with ground-based measures6. Waterwatch include several indicators in their monitoring programs that may serve as early warning indicators of stress to mangroves. These indicators include the quantification of litter production, rates of seedling regeneration, the extent of canopy cover as well as aspects of mangrove structure. the Department of Environment, Water, Heritage and the Arts provides guidelines for State of the Environment reporting.

Photo 9. Research in Avicennia marina mangroves may involve being thigh-deep in black anoxic mud (photo Jon Knight, UQ). The possibility of sinking deep into mangrove mud is ever present.
Existing information and data
Mangroves and coastal saltmarsh of Victoria: Distribution, condition, threats and management comprises the first State-wide assessment of the wetlands that fringe the coast of Victoria. The 514 page report examines the diversity of wetland types and plant communities along the Victorian coast and provides analysis of the ecological condition and major threats to coastal wetlands in Victoria. It also includes the first fine-scale mapping of all current mangrove and saltmarsh wetlands in Victoria.
OzCoasts now delivers mangrove conceptual models (community dynamics, Man-made causes of defoliation, dieback and death, and natural causes of defoliation, dieback and death) for the Queensland Department of Employment, Economic Development and Innovation.
Interactive Habitat Extent and Distribution Mapping Interface. The National Intertidal/Sub-tidal Benthic Habitat Classification scheme (NISB) habitat classes include: mangroves, saltmarsh, seagrass, macroalgae, coral reef, rock-dominated, sediment-dominated and filter feeders (such as sponges). These habitats occur between the approximate position of the highest astronomical tide mark and the location of the outer limit of the photic benthic zone (usually at the 50 to 70 metre depth contour). High spatial resolution polygons with thematic attributes based on NISB are available in the NRM Reporting module, together with national, state and regional summary maps for each habitat.
The OzCoasts database contains mapped areas for a large number of Australian waterways (Figure 1). No differentiation has been made between mangroves and Melaluca stands in OzEstuaries, however this data-set is large and internally consistent. Accurate mapping has been undertaken by a number of state and local governments and academic institutions. For example, Queensland Government – Department of Agriculture & Fisheries(CHRIS) has an interactive map facility that enables users to make detailed vegetation maps for the Queensland coast (see coastal wetland layers) in which mangrove standards are differentiated at the level of genera (Figure 2).
Australia’s Mangroves (by Norm Duke) is an authoritative guidebook on the mangroves of Australian coastal waters. The book has over 500 colour photographs together with clearly illustrated keys, and an innovative water-proof field key in the form of a wheel. There are also distribution maps, fruiting/flowering charts and detailed botanical descriptions.
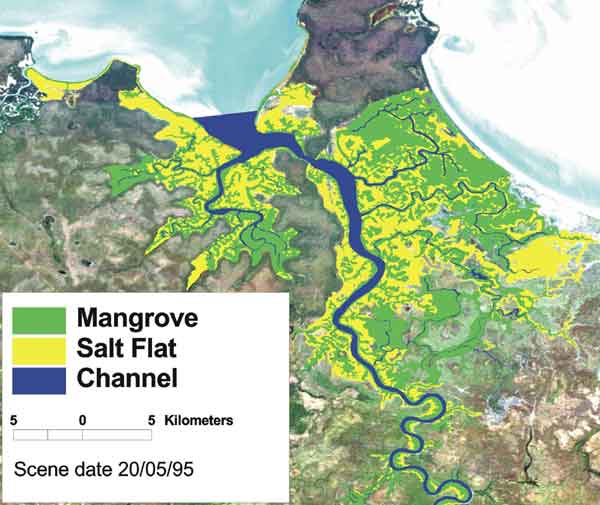
Figure 1. Mangrove areas (green) in the Adelaide River (NT) (from OzEstuaries).
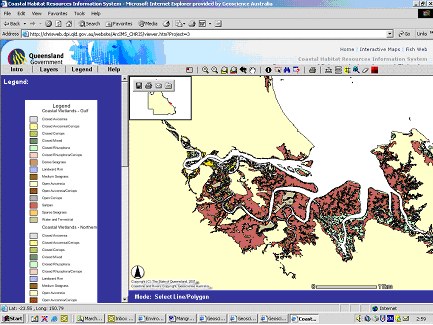
Figure 2. Screen dump showing detailed vegetation map for tidal creeks near the Fitzroy Estuary and Keppel Bay based on CHRIS.
More information on habitat removal/disturbance.
Key questions
- What are the trophic links and exchange of materials between mangroves, and adjacent habitats like salt marshes and seagrasses?2
- What is the relationship between fisheries and mangrove habitats?2
- How can we use measures of mangrove health as bio-indicators of catchment condition?
- How do mangroves adapt to sea level and climate change? Does the rate of change matter?
- How can we use mangroves to stabilise coastal and estuarine shorelines?
Contributors
Mark Breitfuss, Australian School of Environmental Studies, Griffith University
Norm Duke, University of Queensland
Jon Knight, University of Queensland
Kerrylee Rogers, NSW Department of Environment and Conservation
- Duke, N. C. & Watkinson, A. J. (2002). Chlorophyll-deficient propagules of Avicennia marina and apparent longer-term deterioration of mangrove fitness in oil-polluted sediments. Marine Pollution Bulletin (in Press). ↩
- Cappo, M., Alongi, D.M., Williams, D, and Duke, N. 1995. A review and synthesis of Australian Fisheries Habitat Research: Major threats, issues and gaps in knowledge of coastal and marine fisheries habitats. Fisheries Research and Development Corporation. ↩ ↩ ↩ ↩ ↩ ↩
- Duke, N.C. 1992. Mangrove floristics and biogeography. In Tropical Mangrove Ecosystems. Coastal and Estuarine Studies No. 41., (Eds. A.I. Robertson. and D.M. Alongi), American Geophysical Union. ↩
- Heap, A., Bryce, S., Ryan, D., Radke, L., Smith, C., Smith, R., Harris, P. and Heggie, D. 2001. Australian Estuaries & Coastal Waterways: A Geoscience Perspective for Improved and Integrated Resource Management. AGSO Record 2001/07, pp. 118. ↩ ↩
- Fortes, M. 1994. Seagrass resources of ASEAN. In Living Coastal Resources of Southeast Asia: Status and Management, Vol. I, pp 106-112. ↩
- Ward, T., Butler, E. and Hill, B. 1998. Environmental Indicators for National State of the Environment Reporting, Estuaries and the Sea, Commonwealth of Australia, pp. 81. ↩ ↩
- Robertson, A. I. and Phillips, M.J. 1995. Mangroves as filters of shrimp pond effluent: Predictions and biogeochemical research needs. In Proceedings of the Asia-Pacific Symposium on Mangrove Ecosystems, Vol 295, no. 1-3, (Eds Wong, Yuk-Shan and Tam, N.F.Y.), pp. 311-321. ↩
- Harbison, P. 1986. Mangrove muds-A sink or a source for trace metals. Marine Pollution Bulletin 17(6), 246-250. ↩
- Melville, F. and Burchett, M. 2002. Genetic variation in Avicennia marina in three estuaries of Sydney (Australia) and implications for rehabilitation and management. Marine Pollution Bulletin 44, 469-479. ↩
- Volkman, J.K., MIller, G.J., Revill, A.T., and Connell, D.W. 1994. Oil spills. Part 6, In Environmental implications of offshore oil and gas development in Australia, the findings of an independent scientic review (Eds., J.M. Swan, J.M. Neff, and P.C. Young) Australian Petroleum Exploration Association (APEA) & Energy Research and Development Corporation (ERDC), Sydney, PP. 509-695 ↩
- Duke, N.C. 1996. Mangrove reforestation in Panama: an evaluation of planting in areas deforested by a large oil spill. In Restoration of mangrove ecosystems (Ed. C. Field). International Society for Mangrove Ecosystems ISME & International Tropical Timber Organisation ITTO, Okinawa, Japan, pp. 209-232. ↩
- Saintilan, N., Rogers, K. and McKee, K. 2007. Saltmarsh-Mangrove Interactions. In Press ↩
- Ellison, J.C. 1999. Impacts of sediment burial on mangroves. Marine Pollution Bulletin 37, 420-426. ↩ ↩
- Saenger, P. 1988. Gladstone Environmental Survey, Queensland Electricity Commission, Unpublished Final Report. the Department of Environment, Water, Heritage and the Arts. ↩ ↩
- Saintilan, N. and Williams, R.J. 1999. Mangrove transgression into saltmarsh environments in south-east Australia. Global Ecology and Biogeography 8, 117-124. ↩ ↩ ↩
- Conacher, C.A., O’Brien, C.O., Horrocks, J.L. and Kenyon, R.K. 1996. Litter production and accumulation in stressed mangrove communities in the Embley River Estuary, North-eastern Gulf of Carpentaria, Australia. Marine and Freshwater Research 47, 737-743. ↩
- Ross, P. 2002. A view from the things underfoot: the impact of trampling in a temperate mangrove forest. Proceedings of the annual conference of the Australian Marine Sciences Association, 10-12 July 2002, Fremantle WA. ↩
- Rogers, K., Saintilan, N. and Cahoon, D. 2005. Surface elevation dynamics in a regenerating mangrove forest at Homebush Bay, Australia. Wetlands Ecology and Management 13, 587-298. ↩ ↩
- Rogers, K., Wilton, K..M. and Saintilan, N. 2006. Vegetation change and surface elevation dynamics in wetlands of southeast Australia. Estuarine, Coastal and Shelf Science 66, 559-569. ↩
- Vanderzee, M.P. 1988. Changes in saltmarsh vegetation as an early indicator of sea-level rise. In Greenhouse: Planning for climate change (Ed. G.I. Pearmann). Commonwealth Scientific and Industrial Research Organisation (CSIRO), Melbourne, Australia. ↩