Some potential fish indicators
Fishes have long been used as indicators of environmental change because of their generally large size and relative ease of identification1. Different types of fish assemblage indicators include1:
- changes in population abundance i.e. the estimated number of fish of a given species in a pre-defined area;
- the presence/absence of key species;
- modifications of local assemblages including changes in biomass, species composition, ratios of species or feeding groups (e.g. ratios of pelagic to demersal catches2); species diversity and recruitment success (i.e. number of young fish entering a population); and
- measures of health i.e. the frequency of deformities, diseases and other anomalies.
“Fish Stocks” and fishing pressure indicators are also used in State of the Environment (SOE) reporting for Coasts and Oceans (formerly Estuaries and the Sea)3). The fish stocks indicator (e.g. Indicator 4.3 in the Estuaries and the Sea volume) documents the total landed value, total catch weight, a stock assessment and the formal status of the fisheries for the top 25 species in Australia, ranked in terms their dollar values.

Photo 1. School of Silver Drummer. Photo by Euan Harvey (University of Western Australia).
What causes the condition of fish communities to decline?
-
- Eutrophication tends to cause an increase in the biomass ratios of planktivores: demersal fish2. The effects of anoxic & hypoxic events (following from eutrophication) on fish production are hard to predict, as they depend on the balance between the positive and negative effects of nutrient enrichment within the system4. In some cases eutrophication can lead to increased fish densities and greater fish catches. This was the case in the Peel-Harvey Estuary in the 1970s and 1980s.
- Fish kills and general harm to fish communities can occur when dissolved oxygen concentrations drop to lethal levels during the decomposition of organic matter. When oxygen is depleted, anoxic & hypoxic conditions develop and anaerobic organisms take over the degradation of organic matter. Anaerobic respiration gives rise to hydrogen sulfide and ammonia gas which are also toxic to fish5. Rainfall following the dry season in tropical regions can mobilise organic-rich detritus (e.g. rotting weeds, grasses, cane trash and stormwater trash) into coastal waters and this can have a very high biological oxygen demand6.
- Degradation of algal biomass derived from algal blooms can also cause water column de-oxygenation and economic consequences for fisheries production and sales.
- Some point-sources of nutrients to coastal waterways are wastes from aquaculture operations, sewage discharged from yachts, boats & ships and coastal discharges such as outfalls from industry. The risk imposed by point-sources of nutrients in coastal waterways is higher in areas with large population densities or with significant tourism, and can be estimated by the number of point-sources per unit area of coastline. Non-point sources of nutrients from intensive agricultural areas and urban stormwater are often larger and more difficult to control.
- Run-off from acid sulfate soils (ASS) can cause general harm to fish communities and fish kills. The fish die from the metabolic impacts of low pH itself, from lowered dissolved oxygen concentrations (because oxygen is consumed during pyrite oxidation which gives rise to acid sulfate drainage), or from the toxicity of heavy metals mobilised with the drainage. Iron and aluminum in particular play critical roles in fish mortalities7. Sublethal exposure to acid drainage can increase the susceptibility of fish to a cutaneous fungal infection called Red Spot Disease (or Epizootic Ulcerative Syndrome)7. Run-off from acid sulfate soils may result from natural processes but in many cases, the draining of coastal wetlands (e.g. mangroves and salt marshes) leads to the oxidation of pyrite stored in the soil. Coastal waterways with rivers in acid hazard zones are most at risk for acid sulfate drainage.

Photo 2. Often, the most visually obvious effects of acid sulfate soil disturbance are on fish and other aquatic organisms. Run-off transports the acid from acid sulfate soils to local waterways; this exposes aquatic animals to rapid changes in pH, toxic levels of aluminium, iron precipitation, and low dissolved oxygen levels. The photo above illustrates this process, which occurred in south-east Queensland in the Pimpama River. A massive fish kill was associated with the breaking of an extended drought in November 1995 (photo reproduced with permission of ‘QASSIT, Qld Department of Natural Resources and Mines’).
- Introduced pests and diseases are perhaps the most important long-term threat to coastal ecosystems and harvest industries8. This is because exotic species can sometimes spread widely, there are often few prospects for their complete eradication, and the various impacts of exotic species are difficult to predict8. There is evidence that the lesions in Red Spot Disease (see above) are caused by a fungus (Aphanomytces) introduced in the 1970s with the trade of live goldfish9. Coastal waterways with intensive aquaculture or those frequented by ships are presumably most at risk.
- Increased loads of fine sediment to coastal waterways can destroy fish food supplies and disrupt normal reproduction patterns10. Reductions in water clarity from suspended sediment can also cause critical seagrass habitat to contract11. Coastal waterways with highly disturbed catchments (e.g. those in which native vegetation has been removed and replaced by intensive agriculture) are presumably most at risk, especially if the agriculture is on steep slopes. Fish that live in wave-dominated coastal waterways (e.g. deltas, estuaries and strandplains) may be more susceptible to the effects of elevated suspended sediment loads than fish that live in tide-dominated coastal waterways (e.g. deltas, estuaries and tidal creeks). This is because wave-dominated systems have naturally low turbidity levels because tidal currents are generally weak, and because the presence of a barrier at the seaward end protects the waterways from vigorous wave action12. By comparison, tide-dominated coastal waterways are naturally turbid because strong tidal currents cause fine sediment to resuspend12.
- A “critical chain”8 of habitat types is required to maintain a productive and diverse fisheries resource. Coastal habitat (seagrasses, mangroves, salt marshes and other coastal wetlands) is required in the early stages of development of many species of fish9. Estuaries are important nursery areas for certain fish and crustacean species in many regions. There are also a few recreationally and commercially important species such as the cobbler (Cnidoglanis macrocephalus)13 and black bream (Acanthopagrus butcheri)14 which spend the whole of their life cycles in estuaries. A loss or degradation of coastal nursery habitats can lower fish abundance, diversity and recruitment9.
- Changes in environmental flows to coastal waterways impact upon fish communities through the physiological effects of altered salinity, pH and temperature regimes, through interference (or total prevention) of normal patterns of movement, and through changes in the amount of critical habitat and cues for spawning and recruitment9. Coastal waterways with a high impoundment density in the catchment are at risk for reduced freshwater flows. Coastal waterways subject to large volumes of urban stormwater are at risk for increased freshwater flow. Barriers and barrages to tides and river flow can also hinder the movement and migration eggs, larvae and fish, reinforce poor water quality, and provide havens for introduced pest species 915.
- Overfishing and many aspects of harvesting (e.g. trawling, shark-netting, dredging, pocket-netting, angling and marine litter) impact upon fishes9. Such impacts include habitat damage, e.g. by-catching and taxonomic shifts, including increased pelagic:demersal fish biomass ratios and the proliferation of species that capitalise on discarded fish (e.g. birds, fish, mammals and crustaceans)9. “Fishing pressure” can be assessed using two SOE indicators3).
- Risks to fisheries imposed by toxicants (including heavy metals) and pathogenic organisms include diseases, fish kills, bioaccumulation and closures to harvesting9. These concerns tend to be localised to coastal regions with high population densities. The “Industrial Point Source Hazard” and “Volume of Stormwater” indicators can be used to assess toxicant risk from urban and industrial sources. The “Pestcide Hazard” indicator can be used to assess toxicant risk from agricultural sources.
- All of the threats to fish communities listed above are interconnected, and act in synergy. They cannot be treated alone.
Significance of fish communities and their use as indicators
Monitoring of fish communities is advocated as an alternative to water quality monitoring for assessing ecosystem integrity. In general, biotic indices of health are thought superior to water quality programs because they have the potential to record transient events missed by water quality sampling. Fish indices are considered particularly valuable because fish are represented at a variety of trophic levels (including higher-order predators) and their taxonomy is reasonably well known16. In addition, the loss of fish biodiversity can cascade to other aspects of coastal and marine ecosystems where it may manifest in altered stable states, increased incidence of disease, and shifts in dominant species and trophic structure. Impacts to fisheries can also incur severe economic losses17.
Considerations for measurement and interpretation
More research is required documenting the natural temporal and spatial variability in both fish assemblages and their habitats. This natural variability can then be used as a baseline against which human-induced changes can be compared. The increasing use of geographic information systems, and in the case of habitats, remote sensing technology, should help with progress in this area.
The use of remote “video fishing” (with and without bait) offers rapid biodiversity assessment capability, with the option of very precise and accurate length and biomass estimates when stereo-video pairs are used. These video techniques can also be used in combination with other traditional methods to enhance the scope and capability of monitoring programs.
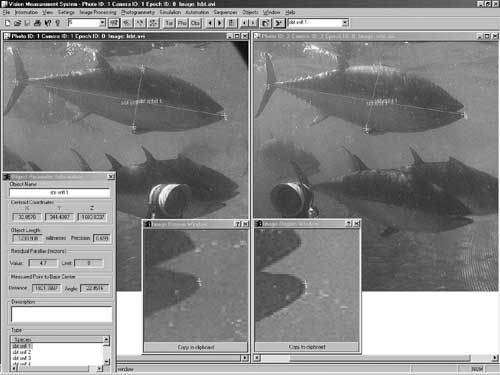
Photo 4. You can stay dry while video fishing for Southern Blue Fin Tuna (photo by Euan Harvey; University of Western Australia).
Existing information and data
Some fish indicator data is held by federal and state agencies and universities. Data on fish abundance, diversity, recruitment and health were sought during the National Land & Water Resources Audit. However, in most cases only species lists or the documentation of the occurrence of important commercial species (e.g. Barramundi) were available. More information on “fish stocks” as indicators (e.g. reporting scales, outputs, analysis and interpretation and data sources) can be found in the Estuaries and the Sea volume of Environmental Indicators for National State of the Environment Reporting3).
More information on biota removal/disturbance.
Contributors
Mike Cappo, AIMS, Queensland
Malcolm Dunning, Department of Primary Industries, Queensland
Euan Harvey, University of Western Australia
Greg Jenkins, Marine and Freshwater Resources Institute, Queenscliff, Victoria
Ian Potter, Centre for Fish and Fisheries Research, Murdoch University
Tom Rose, Waters and Rivers Commission, Western Australia
Glen Young, Centre for Fish and Fisheries Research, Murdoch University
- Stephens, J.S. Jr., Hose, J.E., and Love, M.S. 1988. Fish Assemblages as Indicators of Environmental Change in Nearshore Environments. pp. 91-105 in Soule, D.F. and Kleppel, G.S. (editors), Marine Organisms as Indicators, Springer-Verlag. ↩ ↩
- Caddy, J.F. 2000. Marine catchment basins effects versus impacts of fisheries on semi-enclosed seas. International Council for the Exploration of the Sea – Journal of Marine Science 57, 628-640. ↩ ↩
- Ward, T., Butler, E. and Hill, B. 1998. Environmental Indicators for National State of the Environment Reporting, Estuaries and the Sea, Commonwealth of Australia, pp. 81 (www.ea.gov.au/soe/coasts/estuaries-ind.html) and Australian State of the Environment Committee. 2001. Coasts and Oceans: State of the environment Report 2001. CSIRO Publishing. Collingwood. http://www.ea.gov.au/soe/2001/coasts/pubs/coasts.pdf ↩ ↩ ↩
- Breitburg, D. 2002. Effects of hypoxia, and the balance between hypoxia and enrichment, on coastal fishes and fisheries. Estuaries 25(4b), 767-781. ↩
- Connell, D.W., Miller, G.J. 1984. Chemistry and Ecotoxicology of Pollution. John Wiley & Sons, N.Y. ↩
- Veitch, V. Fish Kills, sunfishqueensland. ↩
- v Sammut, J., Melville, M.D., Callinan, R.B. and Fraser, G.C. 1995. Estuarine acidification: Impacts on aquatic biota of draining acid sulphate soils. Australian Geographical Studies 33(1), 89-100. ↩ ↩
- Cappo, M., Alongi, D.M., Williams, D, and N. Duke. 1995. A review and synthesis of Australian Fisheries Habitat Research: Major threats, issues and gaps in knowledge of coastal and marine fisheries habitats. Fisheries Research and Development Corporation.http://www.aims.gov.au/pages/research/afhr/afhr-00.html ↩ ↩ ↩
- M. Cappo, personal communication, August 2002. ↩ ↩ ↩ ↩ ↩ ↩ ↩ ↩
- Berkman, H.E. and Rabeni, C.F. 1987. Effect of siltation on stream fish communities. Environmental Biology of Fishes 18, 285-294. ↩
- Butler, A. and Jernakoff, P. 1999. Seagrass in Australia: A strategic review and development of an R & D plan. FRDC ↩
- Heap, A., Bryce, S., Ryan, D., Radke, L., Smith, C., Smith, R., Harris, P. and D. Heggie. 2001. Australian Estuaries & Coastal Waterways: A Geoscience Perspective for Improved and Integrated Resource Management. AGSO Record 2001/07, pp. 118. https://data.gov.au/dataset/australian-estuaries-and-coastal-waterways-a-geoscience-perspective-for-improved-and-integrated ↩ ↩
- Laurenson, L.J.B., Neira, F.J. and Potter, I.C. (1993). Reproductive biology and larval morphology of the marine plotosid Cnidoglanis macrocephalus (Teleostei) in a seasonally closed Australian estuary. Hydrobiologia 268 : 179192. ↩
- Sarre, G.A. and Potter, I.C. (1999). Comparisons between the reproductive biology of black bream Acanthopagrus butcheri (Teleostei : Sparidae) in four estuaries with widely differing characteristics. Int. J. Salt Lake Res. 8 : 179-210. ↩
- The Queensland branch of the Australian National Sportfishing Association – http://www.ansaqld.com.au ↩
- Karr, J.R. 1981. Assessment of biotic integrity using fish communities. Fisheries 6(6), 21-27. ↩
- Lenanton, R.C.J. and Potter, I.C. 1987. Contribution of estuaries to commercial fisheries in temperate Western Australia and the concept of estuarine dependence. Estuaries 10(1), 28-35. ↩