What is dissolved oxygen?
Measures of dissolved oxygen (DO) refer to the amount of oxygen contained in water, and define the living conditions for oxygen-requiring (aerobic) aquatic organisms. Oxygen has limited solubility in water, usually ranging from 6 to 14 mg L -11. DO concentrations reflect an equilibrium between oxygen-producing processes (e.g. photosynthesis) and oxygen-consuming processes (e.g. aerobic respiration, nitrification, chemical oxidation), and the rates at which DO is added to and removed from the system by atmospheric exchange (aeration and degassing) and hydrodynamic processes (e.g. accrual/addition from rivers and tides vs. export to ocean)1.
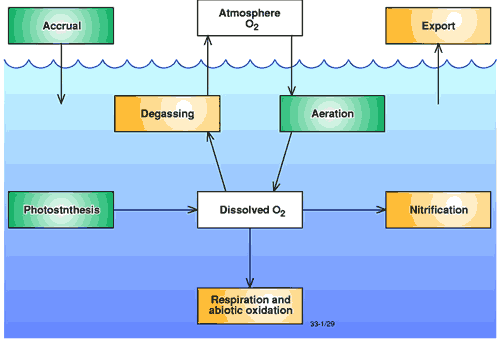
Figure 1. Oxygen dynamics in coastal waters. Processes that increase dissolved oxygen concentrations are shown with green boxes. Processes that decrease dissolved oxygen concentrations are shown with orange boxes. (modified after Connell and Miller, 19841 ).
What causes dissolved oxygen concentrations to change?
- Oxygen solubility varies inversely with salinity, water temperature and atmospheric and hydrostatic pressure.
- Dissolved oxygen consumption and production are influenced by plant and algal biomass, light intensity and water temperature (because they influence photosynthesis), and are subject to diurnal and seasonal variation1.
- DO concentrations naturally vary over a twenty-four hour period due to tidal exchange, and because there is net production of oxygen by plants & algae during the daytime when photosynthesis occurs. By comparison, plants and algae only respire at night time, and this process consumes oxygen. Highly productive systems are expected to have large diurnal DO ranges (see Figure 2).
- Nutrient enrichment stimulates plant and algal growth (and algal blooms) and often results in a mass influx of particulate organic matter to the sediments (eutrophication). The decomposition of this labile organic matter by aerobic microorganisms leads to a rapid acceleration of oxygen consumption, and potential depletion of oxygen in bottom waters.
- Stratification can isolate bottom waters from oxygen enriching processes and can give rise to anoxic & hypoxic events. This problem is most acute in wave-dominated coastal systems (e.g. deltas, estuaries and strandplains and lagoons) because these systems typically have low internal tide penetration. Tides mix the water column and can replenish coastal waterways with oxygen. The baffling effect of seagrass meadows can also impede the mixing process, and maintain bottom water anoxia2.
- Coastal discharges of wastes rich in organic carbon (e.g. from sewage treatment plants, paper manufacturing, food processing and other industries) are produced in large quantities in urban population centres, and can substantially reduce dissolved oxygen concentrations1.
- The oxidation of pyrite found in acid sulfate soils can rapidly strip oxygen from the water, and gives rise to acid drainage. Acid drainage may result from natural processes but in many cases the draining of coastal wetlands (e.g. mangroves and salt marshes) is the cause.
Significance of dissolved oxygen
Most aquatic organisms require oxygen in specified concentration ranges for respiration and efficient metabolism, and DO concentration changes above or below this range can have adverse physiological effects3. Even short-lived anoxic & hypoxic events can cause major “kills” of aquatic organisms. Exposure to low oxygen concentrations can have an immune suppression effect on fish which can elevate their susceptibility to diseases for several years4. Moreover, the toxicity of many toxicants (lead, zinc, copper, cyanide, ammonia, hydrogen sulfide and pentachlorophenol) can double when DO is reduced from 10 to 5 mg L-13. The death of immobile organisms and avoidance of low-oxygen conditions by mobile organisms can also cause changes in the structure and diversity of aquatic communities. In addition, if dissolved oxygen becomes depleted in bottom waters (or sediment), nitrification, and therefore denitrification, may be terminated, and bioavailable orthophosphate and ammonium may be released from the sediment to the water column. These recycled nutrients can give rise to or reinforce algal blooms. Ammonia and hydrogen sulfide gas, also the result of anaerobic respiration, can be toxic to benthic organisms and fish assemblages in high concentrations1.
Considerations for measurement and interpretation
The two standard methods for measuring dissolved oxygen concentrations are membrane electrodes and the Winkler (iodometric) method. Membrane electrodes are the most practical for in situ determinations and for continuous monitoring protocols. The biochemical oxygen demand (or BOD) is another standard procedure used to determine the oxygen requirements of effluents and wastewater. It measures the oxygen utilised during a specified period of organic matter degradation. The solubility of oxygen is affected non-linearly by salinity and water temperature. Therefore, dissolved oxygen measurements are often expressed as percentage saturation values as this parameter is independent of temperature and salinity. Most instrumentation for DO measurement have the facility to convert DO to DO%, when the salinity, temperature and altitude are known. Nomograms to approximate %DO from DO, temperature and altitude data are also available in most standard textbooks on oceanography and limnology.
Spot measurements of DO or %DO are not very useful. The full diurnal range of dissolved oxygen concentrations is required for proper data interpretation, and can be used as an indicator of primary production (see Figure 2). Diurnal DO changes can be tracked over time using moored, continuously recording DO sensors. At the very minimum, a measurement should be taken at midday and dawn to approximate the diurnal range (see Figure 2). Measurements of DO or %DO taken from vertical profiles indicate the extent of mixing or stratification at a monitoring site. The difference between DO and %DO in surface and bottom waters is greatest under stratified conditions. Default trigger values for %DO for different geographic regions in Australia are provided in the ANZECC/ARMCANZ guidelines3, for comparison with median %DO values.
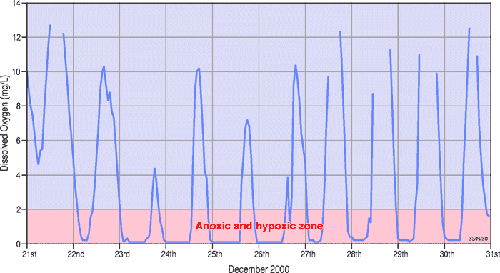
Figure 2. Diurnal variations in surface dissolved oxygen concentrations in Lake Wollumboola in December 2000 (from Haines et al., 20005 ). This lake had a very large diurnal range of dissolved oxygen concentrations at the time of measurement. Concentrations were in the anoxic and hypoxic range at dawn (e.g. 0 – 2 mg L-1) and were in excess of 10 mg L-1 during peak photosynthesis at midday5. Diurnal ranges in dissolved oxygen concentrations of this magnitude are indicative of a very productive lake.
Existing information and data
Major research institutions, universities, consulting companies and government (local and State) agencies gather oxygen data for specific research studies.
Lower limit default trigger values for dissolved oxygen saturation are provided on a bioregional basis in the ANZECC/ARMCANZ Water Quality Guidelines3.
Relationships between nutrient loads and oxygen concentrations in different types of coastal waterways can be explored in the Simple Estuarine Response Models (SERM) developed by CSIRO.
The reference by Best et al. 6, provides dissolved oxygen standards for several Eupoean directives and considers the challenges involved in developing of such standards for the EU Water Framework Directive.
NLWRA 2008: Estuarine, coastal and marine habitat condition, indicator guideline
Indicator guidelines: dissolved oxygen
More information on organic matter (changed from natural) and nutrients (changed from natural).
Contributors
Lynda Radke, Geoscience Australia
- Connell, D.W., and Miller, G.J. 1984. Chemistry and Ecotoxicology of Pollution. John Wiley & Sons, N.Y. ↩ ↩ ↩ ↩ ↩ ↩
- E. McLean 2002. Personal Observation, Coasts and Estuaries Section, LWC Sydney South Coast Region ↩
- ANZECC/ARMCANZ (October 2000) Australian and New Zealand Guidelines for Fresh and Marine Water Quality. ↩ ↩ ↩ ↩
- Mellergaard, S. and Nielson, E. 1987. The influence of oxygen deficiency on the dab population in the eastern North Sea and the southern Kattegat. International Council for the Exploration of the Sea-Collected Papers 1987/E:6: 1-7. ↩
- Haines, P., Skyring G., Stephens, K., Papworth, W. 2001. Managing Lake Wollumboola’s Odour Problem, Proc. 11th NSW Coastal Conference, Newcastle 13-16, November 2001. ↩ ↩
- Best, A.A., Wither, A.W. and Coates, S. 2007. Dissolved oxygen as a physico-chemical supporting element in the water framework directive. Marine Pollution Bulletin 55, 53-64. ↩