What are Diatoms?
Diatoms are single-celled predominantly microscopic algae which consist of two halves (valves) which fit one inside the other to make up a cell (or frustule). Unlike most other algae, diatoms construct their frustule from silica and undergo size reduction during vegetative reproduction since cell division occurs within the confines of the silica cell wall.
Estimates of diatom species diversity vary enormously, predominantly due to conflicting species concepts, with suggested numbers of species ranging from 10,000 to 200,0001. Diatoms account for as much of 25% of the world’s primary production, the majority in the oceans2, and are often the dominant or sub-dominant phytoplankters in Australian estuaries34 and the open ocean5. Species of the toxic diatom Pseudonitzschia have been described from Australian coastal waters though, for the most part, their abundance has been relatively low and populations are mostly non-toxic6.
Careful examination of Australian marine and estuarine taxa67 has revealed that many taxa are shared with the more intensively described flora of the northern hemisphere. However, a number of endemic estuarine species have been described89.
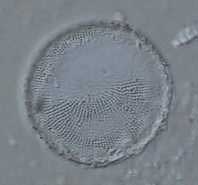
Photo 1. Thalassiosira lacustris: a common brackish water estuarine diatom.
Significance of diatom species composition
Diatoms can be used as indicators of past and present environmental conditions in estuaries. This is because many diatom species have clearly defined, and frequently narrow, preferences for particular habitats (e.g. there are open water-planktonic, plant dwelling-epiphytic, fine sediment-epipelic and sand associated-episammic species) and water quality conditions. In addition, diatoms are abundant and secrete a siliceous cell wall which can preserve in sediment, and which can be used to hind-cast some important aspects of water quality10 and habitat condition11. Changes in diatom species composition along dated sediment cores can thus be highly valuable for determining whether the contemporary conditions of an estuary are within the range of natural variability. Analysis of diatom species assemblages has also led to substantial advances in the understanding of human impact on aquatic habitats and water quality on timescales ranging from sub-decadal to millennial.
For example:
- Changes in the proportion of diatom species in sediment cores has indicated an important change from plant- to plankton-dominated stable states in freshwater lakes on the River Murray floodplain11. Similar hysteresis impacts may have also occurred in estuaries – e.g. a switch from seagrass-dominated ecosystems to phytoplankton-dominated ecosystems – and may be detected by changes in the proportions of planktonic vs. epiphytic (or seagrass associated species) diatom species down sediment cores.
- Changes in the proportion of diatom species in sediment cores have provided important information on the extent and timing of human-induced lake acidification, and also on the recovery of lake systems from acid deposition (see12 for an overview).
Transfer function
Transfer functions derive a quantitative relationship between species composition (in this case diatoms) and water quality parameters (see example in Figure 1). Transfer functions derive numerical optima for each species in a calibration set of sampled sites (typically 30-150) and apply these to species composition data in sediment cores. In this approach, the water quality (e.g. salinity) optimum for a given species is the weighted average of all the salinities where the species occurs, with greatest weight given to those sites where the taxon is most abundant. Estimates of past water quality are derived by multiplying the relative abundance of each fossil taxon by its optimum and summing these values.
There are presently no transfer functions for estuarine systems in Australia. With the development of dedicated coastal lake diatom-water quality transfer functions, it will be possible to estimate some aspects of pre-European water quality in estuaries (see for example13), thereby providing a baseline against which to assess the degree of human impact. Collection of diatom data sets from a variety of localities will also permit statistical matching of pre-impact core samples with those modern diatom samples which are most similar. This process can provide information into which contemporaneous sites best represent pre-impact conditions thus providing important insight into the resilience of coastal waterways.
Diatoms are ideal for biomonitoring where their response to water quality is integrated over periods of days to weeks1014. Hence diatom communities sampled in estuaries may be more informative than “spot” chemistry samples.
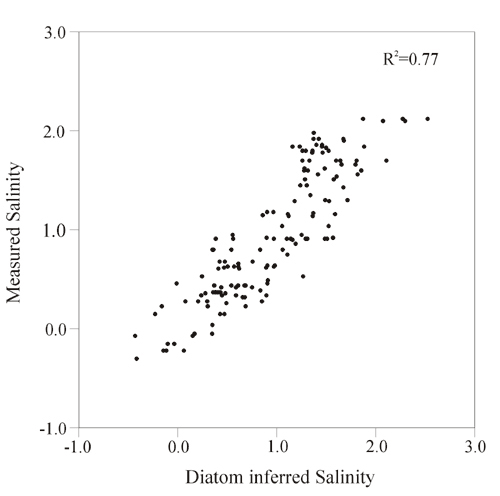
Figure 1. Measured salinity vs. salinity inferred by a diatom transfer function15.
What causes diatom species composition to change?
The most important determinant of diatom species composition in estuarine settings is salinity, and there are a number of schemes16 that outline which diatom species are associated with various salinity levels. The strong association between diatom species composition and salinity has lead to widespread use of diatoms as indicators of past sea level change17, although such techniques have not been applied in Australia. Diatom species composition has also been shown to change in response to:
- pH18;
- total phosphorus (TP)19;
- chlorophyll a concentrations20; and
- water temperature21;
However, at present transfer functions for inferring salinity1522, pH23 and total phosphorus19have only been developed in non-coastal systems in Australia. It is likely that similar models could be developed for estuaries to enhance understanding of the degree and timing of eutrophication, salinity changes (e.g. resulting from river regulation, sea level change and artificial entrance opening) and acidification by acid sulfate drainage24.
Considerations for measurement and interpretation
Species identification in routine enumeration is undertaken using light microscopy. Magnification of at least 750x is required to enable accurate identification of small valves. Differential interference contrast or phase contrast optics are generally employed to allow key features to be highlighted. General valve size and shape, along with the presence/absence and location of a raphe fissure are important for identification of diatoms at generic or higher taxonomic levels. To identify diatoms to species or variety, the arrangement and density of areolae (which themselves form lines known as striae) is one of the most important features25. It is clear that for taxa which are differentiated by small or subtle features that electron microscopy is required to check light microscope identifications6.
Existing information and data
Despite the absence of quantitative models for inferring water quality parameters in Australian estuaries, there have been several studies in which knowledge of diatom ecology has been used to reconstruct environmental histories.
Some interesting findings from these studies were:
- Although salinity at the time of European arrival was at or near its lowest for the period recorded at a number of sites (ranging from 3000-7000 14C years ago, depending on core penetration), salinity declines in response to barrier formation have not been steady. Indeed, in the Tuross system and the lower Snowy, some of the lowest salinities were observed in the period 4000-5000 years before present. In the former system, low salinity has been attributed, using fossil pollen analysis26, to greater catchment wetness, a link also made using particle size data in the Hawkesbury system27.
- artificial entrance opening in the Gippsland Lakes in pre-European times caused a substantial increase in Lake Wellington salinity from < 3 g/L in the pre-European period to values which at times exceed 10 g/L and are regularly > 5 g/L28
- the Snowy River estuary has experienced a substantial increase in salinity since European arrival resulting from the diversion of catchment water29. In Lake Curlip, diatom inferred pre-European salinity was between < 1 and 3 g/L, now it is between 14-20 g/L.
- Diatom records from Lake Alexandrina, SA, indicate that the site has always had naturally high nutrient concentrations, but that further nutrient enrichment and expansion in aquatic plants occurred in the post-European period30.
Contributors
John Tibby, University of Adelaide
- Mann D. and Droop S.J.M. 1996. Biodiversity, biogeography and conservation of diatoms. Hydrobiologia 336, 19-32. ↩
- Milligan A.J. and Morel F.M.M. 2002. A Proton Buffering Role for Silica in Diatoms. Science 297(5588), 1848-1850. ↩
- Chan T. U. and Hamilton D.P. 2001. Effect of freshwater flow on the succession and biomass of phytoplankton in a seasonal estuary. Marine and Freshwater Research 52: 869-84. ↩
- Twomey L., John J. and Thompson P. 2002. Seasonal succession of diatoms and other phytoplankton in a bar built estuary, Wilson Inlet, Western Australia. In: Proceedings of the 15th International Diatom Symposium, Perth, Australia, 28 September – 2 October 1998. J. John (ed.). Königstein, Koeltz Scientific Books: 395-420. ↩
- Hallegraeff, G. M. and Jeffrey, S. W. 1993. Annually recurrent diatom blooms in spring along the New South Wales coast of Australia. Australian Journal of Marine and Freshwater Research 44. 325-34. ↩
- Hallegraeff, G. M. 1994. Species of the Diatom Genus Pseudonitzschia in Australian Waters. Botanica Marina 37, 397-411. ↩ ↩ ↩
- John, J. 1983. The diatom flora of the Swan River Estuary, Western Australia. Bibliotheca Diatomologica 64, 1-360. ↩
- John J. 1991. Climaconeis stromatolitis new species of diatom from Shark Bay, Australia. Diatom Research 6, 49-64. ↩
- John J. 1991. Parlibellus panduriformis, new species (Bacillariophyta) from Shark Bay, Western Australia. Phycologia 30, 556-62. ↩
- Reid M.A., Tibby J.C., Penny D. and Gell P.A. 1995. The Use of Diatoms to Assess Past and Present Water Quality. Australian Journal of Ecology 20(1), 57-64. ↩ ↩
- Reid M., Fluin J., Ogden R., Tibby J. and Kershaw P. 2002. Long-term perspectives on human impacts on floodplain-river ecosystems, Murray-Darling Basin, Australia. Verhandlungen der Internationalen Vereinigung für Theoretische und Angewandte Limnologie 28(2), 710-716. ↩ ↩
- Battarbee R. W., Charles D.F. Dixit, S.S. and Renberg, I. 1999. Diatoms as indicators of surface water acidity. The Diatoms: Applications for the Earth and Environmental Sciences. E. F. Stoermer, J.P. Smol (eds.) New York, Cambridge University Press, 85-127 ↩
- Juggins S. 1992. Diatoms in the Thames Estuary, England: Ecology, palaeoecology, and salinity transfer function. Bibliotheca Diatomologica, 1-216. ↩
- Stevenson, R. J. and Pan, Y. (1999). Assessing environmental conditions in rivers and streams with diatoms. The Diatoms: Applications for the Earth and Environmental Sciences. E. F. Stoermer and J. P. Smol (eds.). Cambridge University Press, New York, 11-40. ↩
- Gell, P.A. 1997. The development of a database for inferring lake salinity, Western Victoria, Australia: towards a quantitative approach for reconstructing past climates. Australian Journal of Botany 45, 389-423. ↩ ↩
- Vos, P. C. and Dewolf, H. 1993. Diatoms as a Tool for Reconstructing Sedimentary Environments in Coastal Wetlands – Methodological Aspects. Hydrobiologia 269/270, 285-296. ↩
- Denys, L. and Baeteman C. 1995. Holocene evolution of relative sea-level and local mean high water spring tides in Belgium – a first assessment. Marine Geology 124, 1-19. ↩
- Hinga, K.R. 2002. Effects of pH on coastal marine phytoplankton, Marine Ecology Progress Series 238, 281-300. ↩
- Tibby J. (in press). Development of a diatom-based model for inferring total phosphorus in south-eastern Australian water storages. Journal of Paleolimnology. ↩ ↩
- Jones, V. J. and Juggins, S. 1995. The construction of a diatom-based chlorophyll-a transfer function and its application at three lakes on Signy Island (maritime Antarctic) subject to differing degrees of nutrient enrichment. Freshwater Biology 34, 433-445. ↩
- Vyverman, W. and Sabbe, K. 1995. Diatom-Temperature Transfer Functions Based on the Altitudinal Zonation of Diatom Assemblages in Papua New Guinea – a Possible Tool in the Reconstruction of Regional Palaeoclimatic Changes. Journal of Paleolimnology 13, 65-77. ↩
- Tibby J. and Reid M. (in press). A model for inferring past conductivity in low salinity waters derived from Murray River diatom plankton. Marine & Freshwater Research. ↩
- Tibby J., Reid M., Fluin J., Hart B.T. and Kershaw A.P. (in press) Assessing long-term pH change in an Australian river catchment using monitoring and palaeolimnological data. Environmental Science and Technology. ↩
- Farago, L. 2002. Using diatoms as bioindicators of past and contemporary water quality condition in acid sulfate environments. In Sustainable Managment of Acid Sulfate Soils, 5th International Acid Sulfate Soils Conference, Twin Towns Services Club, Tweed Heads, NSW Australia, pp. 127-128. ↩
- Sonneman J.A., Sincock A., Fluin J., Reid M., Newall P., Tibby J. and Gell P. (1999). An Illustrated guide to common stream diatoms from temperate Australia. Thurgoona, Cooperative Research Centre for Freshwater Ecology ↩
- Tibby J. 1996. A mid-to late-Holocene diatom and pollen palaeoecology of the Tuross Lake system, South Coast, New South Wales. Monash Publications in Geography, 46, 1-62. ↩
- Devoy R.J., Dodson J.R., Thom B.G. and Nichol S. 1994. Holocene environments in the Hawkesbury Valley, New South Wales – a comparison of terrestrial and marine records. Quaternary Science Reviews 13(3), 241-256. ↩
- Reid, M., 1998. A fine resolution palaeoecology of recent sediments from Lake Wellington, Gippsland Lakes, Victoria. In: Proceedings of the First Australian Diatom Workshop, Warrnambool, Australia, 1-3 February, 1997. P. Newall (ed.) Deakin University School of Ecology and Environment, Technical Paper 1998/1, 86-91. ↩
- MacGregor A. 2002. A palaeo-ecological reconstruction of the Lower Snowy River, East Gippsland, Victoria: environmental response to climate change, land use and river regulation. B.Env (Hons) thesis, Department of Geographical and Environmental Studies, University of Adelaide. ↩
- Fluin, J. 2001. A diatom-based palaeolimnological investigation of the lower Murray River (south east Australia). Unpublished PhD thesis, School of Geography and Environmental Science, Monash University. ↩