What is chlorophyll a?
Chlorophyll a is a green pigment found in plants. It absorbs sunlight and converts it to sugar during photosynthesis. Chlorophyll a concentrations are an indicator of phytoplankton abundance and biomass in coastal and estuarine waters. They can be an effective measure of trophic status1, are potential indicators of maximum photosynthetic rate (P-max)2 and are a commonly used measure of water quality. High levels often indicate poor water quality and low levels often suggest good conditions. However, elevated chlorophyll a concentrations are not necessarily a bad thing. It is the long-term persistence of elevated levels that is a problem. For this reason, annual median chlorophyll a concentrations in a waterway are an important indicator in State of the Environment3.
What causes elevated chlorophyll concentrations to change?
It is natural for chlorophyll a levels to fluctuate over time. Chlorophyll a concentrations are often higher after rainfall, particularly if the rain has flushed nutrients into the water. Higher chlorophyll a levels are also common during the summer months when water temperatures and light levels.
Tidal regime is an important control on algal biomass (see Figure 2)4. Strong tidal mixing lowers chlorophyll a concentrations because the residence time of algae in the photic zone is reduced4. Tidal mixing also causes fine sediment to resuspend, and the elevated turbidity levels that result reduce the amount of light available for photosynthesis.
Changes to systems which decrease (e.g. construction of canal estates) or increase (e.g. breakwaters, training water and dredging) flushing rates influence chlorophyll a concentrations because flushing dilutes nutrients and moves them away from plants, making them less available. Conversely, slow moving or stagnant waters let nutrients increase and cell numbers grow.
Elevated concentrations of chlorophyll a can reflect an increase in nutrient loads (see Figure 1) and increasing trends can indicate eutrophication of aquatic ecosystems.
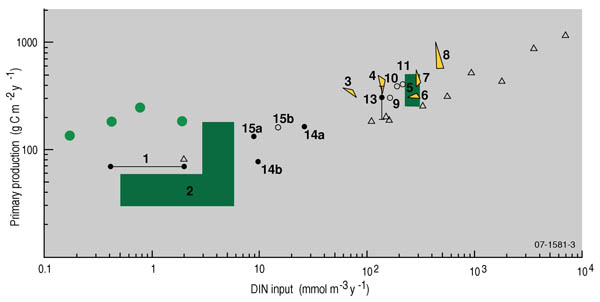
Figure 1. Annual inorganic nitrogen loads vs. primary production for some marine and coastal waterways from around the world (from Nixon 19925; reproduced with permission of Scott Nixon). Data indicated by lime green dots are based on the work of Samantha Wellman (Ph.D in prep), and are reproduced with the permission of P. Scanes (NSW Department of Environment and conservation).
Significance of elevated chlorophyll a concentrations
Phytoplankton productivity is one of the main forces regulating our planetary climate for via impacts on atmospheric carbon dioxide levels which are closely linked to the oceanic carbon dioxide concentrations6. However, excessive water column productivity, expressed by high chlorophyll a concentrations, can supply large amounts of easily decomposition (i.e. labile) organic matter to the sediments. The decomposition of algal biomass can increase the diurnal amplitude of water column pH and dissolved oxygen fluctuations, and in some cases may lead to anoxic & hypoxic events. Moreover, elevated chlorophyll a levels indicate high numbers of phytoplankton and free floating macroalgae which can shade seagrass meadows leading to a decline in seagrass distribution7. The above changes can translate into changes in animal and plant species diversity8.
Waterways susceptible to elevated chlorophyll concentrations
Nutrient impacts on coastal waterways (reflected in elevated chlorophyll a concentrations) depends on both the loads and bioavailability of nutrients, and the extent to which hydrodynamic features (e.g. residence times and mixing) and turbidity levels buffer nutrient loads1.
Chlorophyll a concentrations are generally low in meso- and macro-tidal systems (e.g. deltas, estuaries and tidal creeks with mean tidal range >2m) due to the strong tidal flushing, and because tidal resuspension gives rise to high turbidity levels (discussed above) (Figure 2). By comparison, micro-tidal coastal waterways (e.g. wave-dominated estuaries, enclosed bays and shallow coastal lagoons with mean tidal range nutrients information sheet).
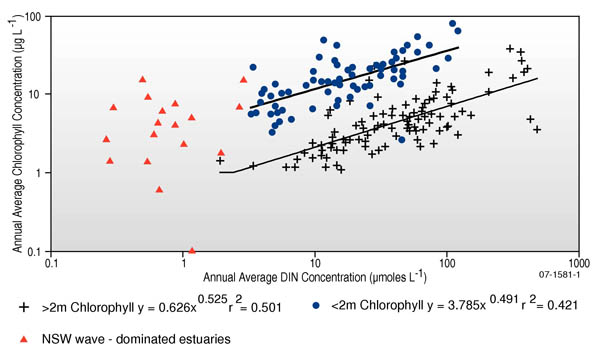
Figure 2. Annual average chlorophyll-a concentrations vs annual average dissolved inorganic nitrogen (DIN) concentrations for some non-Australian micro-tidal (tidal range 2 m) (data from Monbet (1992) 4 ) and some NSW micro-tidal estuaries (data from 9). (Image reproduced with permission of K. Hinga10 and P. Scanes (NSW Department of Environment & Conservation).
Advantages of measuring chlorophyll a over nutrient concentrations
Integration over time
Phytoplankton assimilate available nutrients over their lifetime, whereas water column inorganic nutrient concentrations are notoriously variable over much shorter time scales6.
Sensitivity
Phytoplankton respond rapidly to pulsed nutrient inputs that might otherwise go undetected by regular nutrient sampling6.
Ease of collection
Chlorophyll a samples require minimal processing and storage in the field and are not easily contaminated6.
Cost
Chlorophyll a is cheap in comparison to the analysis of a full suite of dissolved nutrients6.
Detect ability
Remote sensing can detect chlorophyll concentrations but cannot detect nutrient concentrations such as phosphorous and nitrogen directly6.
Considerations for measurement and interpretation
Interpretation of chlorophyll a data
Phytoplankton populations can exhibit significant spatial and temporal variation (e.g. some algae migrate up and down within the water column) and concentrations can be quite different along lengths of the waterways or estuaries. The design of field studies needs to take this into account. However, surface chlorophyll a concentrations discussed below) can be estimated from satellite data, and can redress this gap in our understanding of the temporal and spatial variations of phytoplankton biomass
There is not always a clear relationship between chlorophyll concentration and biomass or cell number because of species differences in size and chlorophyll content1.
Measurement of chlorophyll a
Chlorophyll a is usually collected from water samples of a known volume that are filtered through fine mesh filter paper (0.45 micron) which is then analysed for chlorophyll a concentrations. There are three standard methods for determining chlorophyll a concentrations: spectrophotometry, fluorometry and high performance liquid chromatography (HPLC). Spectrophotometry is the most commonly used method, although the slower and more demanding HPLC method is able to differentiate between chlorophyll types and accessory pigments. Fluorometers transmit an excitation beam of light in the blue range (440 nm – 460 nm) and detect the light fluoresced by chlorophyll in a sample in the red wavelength (685 nm). This fluorescence is directly proportional to the concentration of chlorophyll. Although fluorescence is an indirect method for measuring chlorophyll a, field instrumentation allows this measurement to be made in situ and in a time-series.
Remote Sensing of chlorophyll a
Satellite remote sensing of chlorophyll concentrations rely on the absorption and scattering characteristics of phytoplankton and the way these optical properties affect the underwater light field and the reflectance (Rrs) or radiance (nLw) values measured by the satellite sensor6. Information on chlorophyll a concentrations is obtained by using appropriate algorithms that relate measurements of remote sensing reflectance spectra either directly to chlorophyll concentration (empirical algorithms), or to optical properties of phytoplankton and other optically active materials in the water (semi-analytical algorithms, based on radiative transfer and theoretical relationships) (See Figure 3; 6).
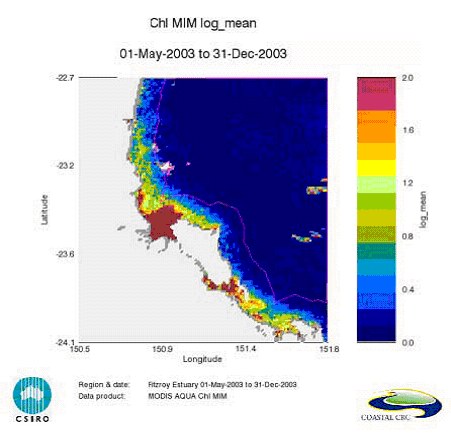
Figure 3. Map of the log-transformed chlorophyll values (log mean) for the dry season in Keppel Bay, Queensland, for the period May – December 2003 of the CHL retrieval by the MIM algorithm [from6].
Existing information and data
Various data sets can be found in state agencies, Commonwealth institutions and universities. Further details can be obtained on eutrophication and algal blooms from the National Eutrophication Management Program. The ANZECC/ARMCANZ Water Quality Guidelines1 list default trigger values for chlorophyll a concentrations for coastal waterways in different geographic regions. The chlorophyll a threshold for impacts on fish is generally considered to be 100 micrograms of chlorophyll a per litre8. the Department of Environment, Water, Heritage and the Arts provides guidelines for State of the Environment reporting3. The relationships between chlorophyll a concentrations and nutrient loads in different types of coastal waterways can be explored using the Simple Estuarine Response Model developed by CSIRO.
More information on nutrients (changed from natural).
Research needs and questions
Separate sets of default trigger values and water quality targets should be derived for chlorophyll a concentrations in tide- and wave-dominated systems (see Figure 2).
NLWRA 2008: Estuarine, coastal and marine habitat condition, indicator guideline
Surface water chlorophyll a concentration
Contributors
Vittorio Brando, CSIRO Land and Water
Andrew Moss, Queensland EPA
Lynda Radke, Geoscience Australia
David Rissik, NSW Land and Water Conservation
Tom Rose, Waters and Rivers Commission, Western Australia
Peter Scanes, Department of Environment and conservation, NSW
Samantha Wellman, School of Applied Sciences, University of Newcastle
- ANZECC/ARMCANZ (October 2000) Australian and New Zealand Guidelines for Fresh and Marine Water Quality. ↩ ↩ ↩ ↩
- Wellman, S., Redden, A., Rissik, D., and Scanes, P. 2002. Phytoplankton production in near-pristine and urban-influenced coastal lagoons,Proceedings of the annual conference of the Australian Marine Sciences Association, 10-12 July 2002, Fremantle, WA. ↩
- Ward, T., Butler, E. and Hill, B. 1998. Environmental Indicators for National State of the Environment Reporting, Estuaries and the Sea, Commonwealth of Australia, pp. 81. ↩ ↩
- Monbet, Y. 1992. Control of phytoplankton biomass in estuaries: A comparative analysis of microtidal and macrotidal estuaries. Estuaries 15(4), 563-571. ↩ ↩ ↩
- Nixon, S.W. 1992. Quantifying the relationship between nitrogen input and the productivity of marine ecosystems. Advanced Marine Technology Conference, No. 5, Japan, pp. 57-83. ↩
- Brando, V., Dekker, A., Marks, A., Qin, Y and Oubelkheir, K. 2006. Chlorophyll and suspended sediment assessment in a macrotidal tropical estuary adjacent to the Great Barrier Reef: spatial and temporal assessment using remote sensing. Cooperative Research Centre for Coastal Zone, Estuary & Waterway Management Technical Report 74. ↩ ↩ ↩ ↩ ↩ ↩ ↩ ↩ ↩
- Duarte, C.M. 1995. Submerged aquatic vegetation in relation to different nutrient regimes. Ophelia 41, 87-112. ↩
- Nielson, J. and P. Jernakoff, P. 1996. A review of the interaction of sediment and water quality with benthic communities. Port Phillip Bay Environmental Study. Technical Report No. 25, 1-130. ↩ ↩
- Scanes P., G. Coade, M. Doherty and R. Hill, 2007, Evaluation of the utility of water quality based indicators of the estuarine lagoon condition in NSW, Australia. Est. Coast. Shelf Sci. 74: 306-319. ↩
- Hinga, K.R., Jeon, H. and Lewis, N.F. 1995. Marine eutrohication review I: Quantifying the effects of nitrogen enrichment on phytoplankton in coastal ecosystems. NOAA Coastal Ocean Office, Silver Spring, MD, 36p. ↩