What are anoxic and hypoxic events?
Dissolved oxygen depletion in coastal and estuarine waters is a growing global concern. Bottom water dissolved oxygen (DO) concentrations are near zero under anoxic conditions, whereas they are <2.0 mg L-1 O2 under hypoxic conditions.
The incidence of anoxic and hypoxic events is a suggested water quality indicator in the ANZECC/ARMCANZ water quality guidelines1, and was adopted as an indicator of ecosystem integrity during the National Land and Water Resources Audit.
What causes anoxic and hypoxic events?
Anoxic and hypoxic events are caused by the decomposition of organic matter by oxygen-utilising bacteria. In many cases, anoxia and hypoxia result from eutrophication (e.g. enhanced sedimentation of POM to bottom waters) and reflect the underlying problem of excessive nutrient loads.
Some point-sources of nutrients to coastal waterways are coastal discharges, including outfalls from industry, wastes from aquaculture operations and sewage discharged from yachts, boats & ships. The risk imposed by point-sources of nutrients in coastal waterways is higher in areas with large population densities or with a significant tourism, and can be estimated by the number of point-sources per unit area of coastline. Non-point sources of nutrients from intensive agricultural in catchments and urban stormwater are often larger and more difficult to control.
Rainfall following the dry season in tropical regions can also mobilise organic-rich detritus (e.grotting weeds, grasses, cane trash and stormwater trash) into coastal waters and these can have a very high biological oxygen demand2.
Waterways susceptible to anoxic and hypoxic events
Severe DO depletion in the coastal zone is usually associated with density stratification in wave-dominated coastal systems (e.g. deltas, estuaries and strandplains and coastal lagoons) characterised by low levels of tidal mixing, and subject to elevated nutrient loads. Density stratification occurs when freshwater flows seaward over denser and more saline marine water (Figure 1), and is therefore intensified after periods of heavy rainfall. Temperature stratification can also occur – most commonly in systems such as coastal lagoons and strandplains – and results from solar warming of surface waters. In both cases, stratification restricts vertical mixing and therefore the replenishment of bottom water with oxygen derived from the atmosphere. Severe oxygen depletion is not likely to be a significant issue in tide-dominated coastal systems (e.g. deltas, estuaries and tidal creeks), because tidal mixing reduces the potential for stratification (Figure 2).
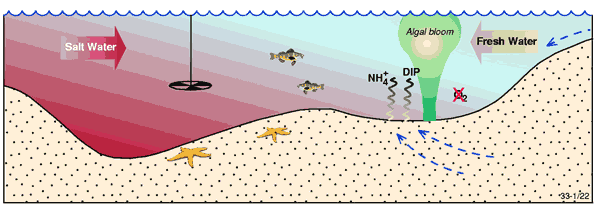
Figure 1. Conceptual model illustrating oxygen depletion and nutrient regeneration in bottom waters (and sediments) in the stratified portion of an estuary e.g. where saline water (red) underlies riverine water (blue).

Figure 2. Mixing is generally low in wave-dominated coastal systems and naturally high in tide-dominated coastal systems (from Heap et al., 20013 ).
Significance of anoxic and hypoxic events
A reduction in dissolved oxygen concentrations is amongst the most important effects of eutrophication on aquatic organisms4. Hypoxia can cause direct mortality, reduced growth rates and altered behaviour and distributions of fish4 and other organisms. In addition, bottom-water hypoxia can interact with elevated water temperatures at the surface to produce a “temperature-oxygen squeeze” effect, which can greatly reduce the amount of summer habitat available for some species5. Eggs and larvae of fish (and crustaceans) may be particularly susceptible to this effect because these life history stages are less able to avoid unfavourable conditions, and because they live in near shore areas, such as estuaries, where too-high water temperatures and too-low oxygen conditions often occur6. Changes in fish assemblages and crustaceans in response to hypoxia and & anoxia can render these organisms more susceptible to fishing pressure, and can increase the abundance of non-targeted species in by-catch4.
Dissolved oxygen status also influences the uptake or release of nutrients from sediment. When oxygen is depleted, the nitrification pathway is blocked, and denitrification efficiencies may be lowered. As a consequence, more nutrients (e.g. nitrogen and phosphorous) are released from the sediment in bio-available forms7. These nutrients help to sustain algal blooms, and therefore continue the supply organic matter to the sediments7. With organic matter (energy) diverted from invertebrate consumption to microbial decomposition, the natural pattern of energy flow is altered, and pelagic and opportunistic species are favoured8. Indeed, an increased ratio of planktivore:demersal fish biomass is an important effect of eutrophication9. Low bottom water oxygen concentrations are also conducive to the build-up of toxic compounds such as hydrogen sulfide and ammonia gas, which can also be harmful to benthic organisms and fish. Even short-lived anoxic events can cause the mass mortality of fish and benthic organisms10.
Overall, anoxic and hypoxic events can cause large reductions in the abundance, diversity and harvest of fish in affected waters4, and can contribute to an overall loss of bio-diversity8. However, the extent to which bottom water anoxia causes declines in overall fish production depends on a balanced between the negative and positive and effects of eutrophication in the full spectrum of habitats within the system4.
Considerations for measurement and interpretation
Anoxic and hypoxic events are often recognised by the deleterious effects they have on aquatic ecosystems (e.g. fish kills). As such, by the time such events are detected, substantial damage may have already occurred to the ecosystem. Ideally, the spatial and temporal extent of anoxia, the response of benthic communities and impacts on fisheries stocks should be documented8. Biological and chemical indices from dated sediment records are useful for documenting changes in the magnitude and frequency of oxygen stress over time, and as to whether such changes result from human or natural causes7.
Remediation
Properly designed hypo-limnetic aeration and oxygenation systems can replenish dissolved oxygen in waterways while preserving stratification. The three primary devices are Speece Cone, airlift aerator and bubble-plume diffuser, and these are extensively reviewed in11. Artificial oxygenation (diffuser-type) has been used with success in the Canning River in Perth in Western Australia, as part of the Swan-Canning Cleanup Program. A scientific manuscript based on this work highlighted large drops in ammonium and total phosphorus concentrations when the oxygenation plant was operating, as well as an increase in nitrifying bacteria12.
Existing information and data
Major research institutions, universities and government (local and State) agencies gather oxygen data for specific research studies. Some information on anoxic and hypoxic events in Australian coastal waterways was compiled during the National Land & Water Resources Audit. In most cases, no data was available. However, localised or short-lived periods of hypoxia were reported in the Derwent and Huon estuaries (TAS) and in the Tuggerah Lakes (NSW). Prolonged and extensive anoxia is experienced in the Gippsland Lakes.
More information on organic matter (changed from natural) and nutrients (changed from natural).
Key questions
A key research question is “what combinations of nutrient loadings and hydrodynamic regimes cause anoxic/hypoxic conditions in estuaries and coastal waterways?” 4. Questions such as these are perhaps best addressed through ecosystem modelling studies (see for example The Gippsland Lakes Environmental Study), and can be explored by managers using the Simple Estuarine Response Models SERM (www.per.marine.csiro.au/serm/index.htm) developed by CSIRO.
Contributors
Lynda Radke, Geoscience Australia
- ANZECC/ARMCANZ (October 2000) Australian and New Zealand Guidelines for Fresh and Marine Water Quality. (www.ea.gov.au/water/quality/nwqms/#quality) ↩
- Veitch, V. Fish Kills, sunfishqueensland. ↩
- Heap, A., Bryce, S., Ryand, D., Radke, L., Smith, C., Smith, R., Harris, P. and D. Heggie. 2001. Australian Estuaries & Coastal Waterways: A Geoscience Perspective for Improved and Integrated Resource Management. AGSO Record 2001/07, pp. 118. Organisations/Geoscience Australia/Australian Estuaries and … ↩
- Breitburg, D. 2002. Effects of hypoxia, and the balance between hypoxia and enrichment, on coastal fishes and fisheries. Estuaries 25(4b), 767-781. ↩ ↩ ↩ ↩ ↩ ↩
- Coutant, C.C. 1985. Striped bass, temperature, and dissolved oxygen: A speculative hypothesis for environmental risk. Transactions of the American Fisheries Society 114, 31-61. ↩
- Hypoxia. The Ecological Society of America (https://www.esa.org/esa/wp-content/uploads/2012/12/hypoxia.pdf) ↩
- Heggie, D.T., Skyring, G.W., Orchardo, J., Longmore, A.R., Nicholson, G.J., and Berelson, W.M. 1999. Denitrification and denitrifying efficiencies in sediments of Port Phillip Bay: direct determinations of biogenic N2 and N-metabolite fluxes with implications for water quality. Marine and Freshwater Research 50, 589-596. ↩ ↩ ↩
- Nelson, T.A., Blackwelder, P., Hood, T., McKee, B., Romer, N., Alvarez-Zarikian, C., Romer, N., and Metz, S. 1994. Time-based correlation of biogenic, lithogenic and authigenic inputs in the Gulf of Mexico NECOP study area. Estuaries 17(4), 873-885. ↩ ↩ ↩
- Caddy, J.F. 2000. Marine catchment basins effects versus impacts of fisheries on semi-enclosed seas. In International Council for the Exploration of the Sea – Journal of Marine Science 57, 628-640. ↩
- Stachowitsch, M. 1992. Benthic communities: eutrophication’s “memory mode”. In Marine Coastal Eutrophication (Ed., Vollenwieder, R.A., Marchetti, R and Vivani, R. Elsevier, Amsterdam , London, New York, Tokyo, pp. 1017-1028. ↩
- Singleton, V.L. and Little, J.C. in press. Designing hypolimnetic and oxygenation systems – a review. Environmental Science and Technology. ↩
- Greenop, B., Lovatt, K., Robb, M. 2001. The use of artificial oxygenation to reduce nutrient availability in the Canning River, Western Australia. Water Science and Technology 43(9), 133-144. ↩